Our eosinophilosophy
eos counts in
severe eosinophilic asthma
Severe eosinophilic asthma (SEA) is a distinct phenotype of severe asthma. The SEA phenotype can include airway inflammation, increased eosinophil levels, poor asthma control, and increased rate of exacerbations.1
Half of severe asthma patients have persistent airway tissue eosinophils resulting in poor prognosis and frequent exacerbations.2
Eosinophilic airway inflammation in SEA3
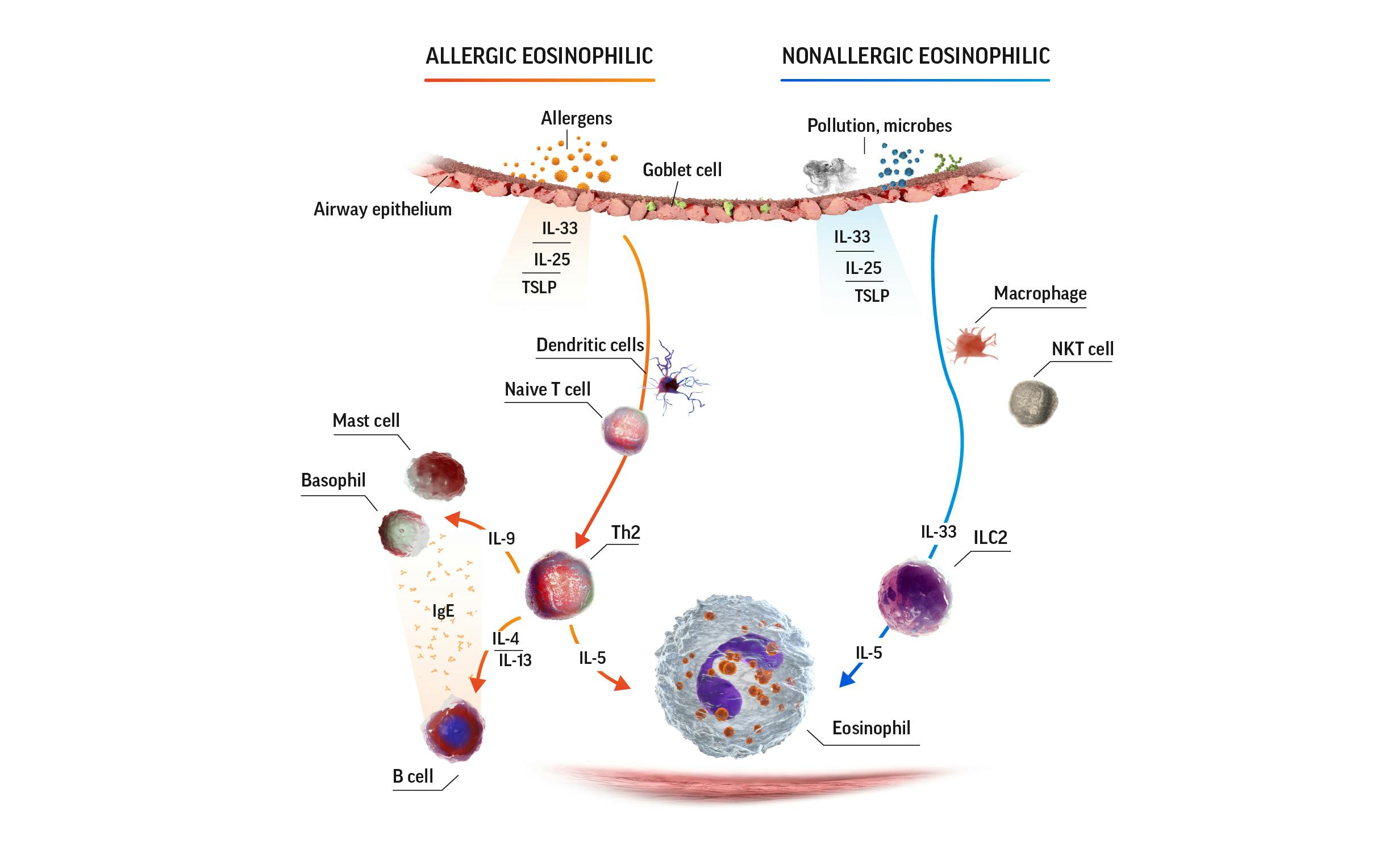
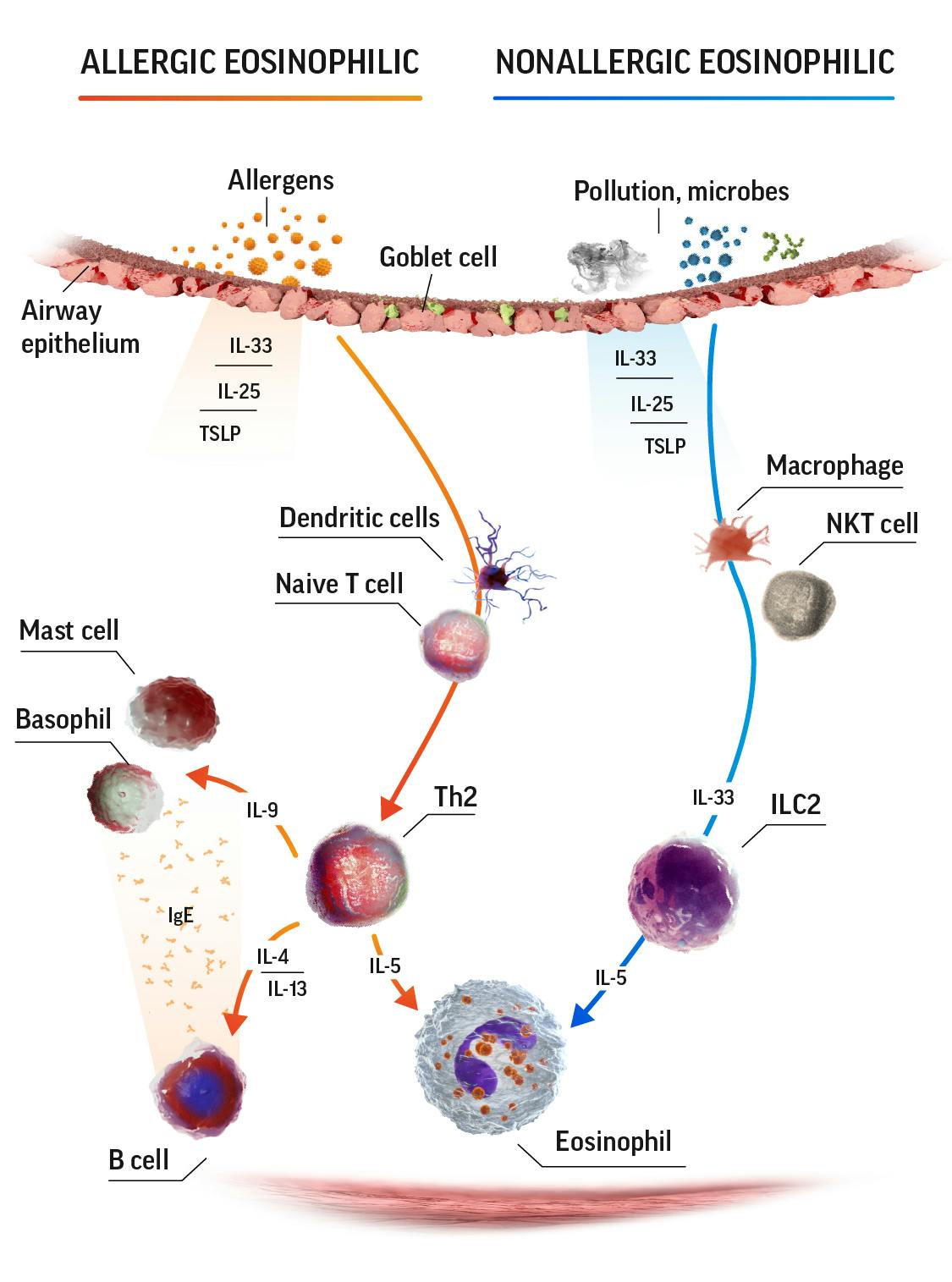
IgE=immunoglobulin E; IL=interleukin; ILC2=group 2 innate lymphoid cells; NKT=natural killer T; Th2=T helper cell type 2; TSLP=thymic stromal lymphopoietin.
Eosinophils release a range of cytokines and chemokines that contribute to the pathophysiology of asthma, including4-8:
IL-4
- Macrophage polarization
IL-13
- Macrophage polarization
- Basophil activation
- Contributes to mucus production
- Contributes to airway hyperresponsiveness
IL-5
- Responsible for maturation, activation, and survival of eosinophils
Transformation growth factor beta (TGF-β)
- Promotes airway remodeling
Leukotrienes
- Contributes to airway hyper-responsiveness
Major basic protein (MBP)
- Stimulates mast cells and basophils
- Causes tissue damage by stimulating tissue-residing epithelial cells to express profibrotic mediators
- Stimulates IL-8 secretion from neutrophils which can lead to neutrophilia
Eosinophil peroxidase (EPO)
- Stimulates mast cells
- Causes tissue damage by stimulating tissue-residing epithelial cells to express profibrotic mediators
- Induces platelet aggregation
Eosinophils in SEA increase the risk of exacerbations
The chronic inflammation caused by eosinophils may result in harmful structural changes to the airway walls, known as remodeling, which leads to bronchial wall thickening and reduced air flow. This inflammation could also cause damage resulting in airway smooth muscle contraction, loss of lung function, and airway hyperresponsiveness.9 Remodeling can also increase the frequency and worsen the severity of exacerbations.1
Elevated EOS are associated with asthma exacerbations10,11
Patients with asthma and blood eosinophils ≥300 cells/μL and fractional exhaled nitric oxide (FeNO) >50 ppb were nearly 4x more likely to have a severe exacerbation than those with lower blood eosinophils (<300 μL) and low FeNO (<25 ppb).10 Intermediate and high blood eosinophils were associated with asthma-related visits to the emergency department.11
Could it be EGPA?
Asthma and elevated eosinophils also occur in patients with eosinophilic granulomatosis with polyangiitis (EGPA).14 See the criteria for EGPA to know the additional signs to look for.
Eosinophil counts can help guide SEA treatment decisions
Elevated eosinophils are an indicator of a type of underlying inflammation called Type 2 inflammation. This type of inflammation in severe asthma may be relatively refractory to high-dose inhaled corticosteroids (ICS).1,13,15 Although the inflammation may respond to oral corticosteroids (OCS), there are risks of developing adverse effects.1,13 Knowing if a patient with severe asthma has elevated eosinophils can therefore guide decisions in determining appropriate treatments.
Cardiovascular/cerebrovascular disease
Cataracts
Depression/anxiety
Gastrointestinal ulcer/bleeds
Hypertension
Infection
Osteoporosis/fracture
Renal impairment
Sleep disturbance
Type 2 diabetes
Weight gain/obesity
Targeting eosinophil levels and inflammation could be critical to managing SEA. Eosinophil levels can help identify the severe eosinophilic/Type 2 inflammation phenotype in patients on high-dose ICS or daily OCS, using the following criteria13:
- Blood eosinophils levels of ≥150 cells/µL, and/or
- FeNO ≥20 ppb, and/or
- Sputum eosinophils ≥2%
In addition, blood eosinophil levels of ≥150 or ≥300 cells/µL or FeNO ≥25 ppb are also often used by payers to determine biologic therapy eligibility and higher blood eosinophils or FeNO are potentially predictive of good responsiveness to different biologic therapies.13
It is important to note that ~65% of patients on medium-to-high-dose ICS-long-acting beta2-agonist shift their eosinophil levels over a period of 12 months, therefore, additional assessments in patients with low blood eosinophil levels have been recommended. Additionally, OCS can suppress eosinophil levels, and therefore assessments should be made either before OCS start or at the lowest possible OCS dose.13
Knowing your severe asthma patients' eosinophil count can help you give them a more targeted treatment13
References:
- Chung KF, Wenzel SE, Brozek JL, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma [published corrections appear in Eur Respir J. 2014;43(4):1216 and Eur Respir J. 2018;52(1):1352020.] Eur Respir J. 2014;43(2):343-373.
- Buhl R, Humbert M, Bjermer L, et al. Severe eosinophilic asthma: a roadmap to consensus. Eur Respir J. 2017;49(5):1700634.
- Brusselle G, Bracke K. Targeting immune pathways for therapy in asthma and chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2014;11 Suppl 5:S322-S328.
- Varricchi G, Senna G, Loffredo S, Bagnasco D, Ferrando M, Canonica GW. Reslizumab and eosinophilic asthma: one step closer to precision medicine? Front Immunol. 2017;8:242.
- Ramirez GA, Yacoub MR, Ripa M, et al. Eosinophils from physiology to disease: a comprehensive review. Biomed Res Int. 2018;2018:9095275.
- Kouro T, Takatsu K. IL-5- and eosinophil-mediated inflammation: from discovery to therapy. Int Immunol. 2009;21(12):1303-1309.
- McBrien CN, Menzies-Gow A. The biology of eosinophils and their role in asthma. Front Med (Lausanne). 2017;4:93.
- Wen T, Rothenberg ME. The regulatory function of eosinophils. Microbiol Spectr. 2016;4(5):10.1128/microbiolspec.MCHD-0020-2015.
- Trivedi SG, Lloyd CM. Eosinophils in the pathogenesis of allergic airways disease. Cell Mol Life Sci. 2007;64(10):1269-1289.
- Price DB, Bosnic-Anticevich S, Pavord ID, et al. Association of elevated fractional exhaled nitric oxide concentration and blood eosinophil count with severe asthma exacerbations. Clin Transl Allergy. 2019;9:41.
- Malinovschi A, Fonseca JA, Jacinto T, Alving K, Janson C. Exhaled nitric oxide levels and blood eosinophil counts independently associate with wheeze and asthma events in National Health and Nutrition Examination Survey subjects. J Allergy Clin Immunol. 2013;132(4):821-827.e1-5.
- de Groot JC, ten Brinke A, Bel EH. Management of the patient with eosinophilic asthma: a new era begins [published correction appears in ERJ Open Res. 2016;2(3):0024-2015-ERR]. ERJ Open Res. 2015;1(1):00024-2015.
- Global Initiative for Asthma (GINA). Global strategy for asthma management and prevention. 2024 main report. https://ginasthma.org/2024-report/. Accessed July 25, 2024.
- Baldini C, Talarico R, Della Rossa A, Bombardieri S. Clinical manifestations and treatment of Churg-Strauss syndrome. Rheum Dis Clin North Am. 2010;36(3):527-543.
- Wenzel SE, Schwartz LB, Langmack EL, et al. Evidence that severe asthma can be divided pathologically into two inflammatory subtypes with distinct physiologic and clinical characteristics. Am J Respir Crit Care Med. 1999;160(3):1001–1008.
- Sullivan PW, Ghushchyan VH, Globe G, Schatz M. Oral corticosteroid exposure and adverse effects in asthmatic patients. J Allergy Clin Immunol. 2018;141(1):110-116.e7.
- Price DB, Trudo F, Voorham J, et al. Adverse outcomes from initiation of systemic corticosteroids for asthma: long-term observational study. J Asthma Allergy. 2018:11:193-204.
